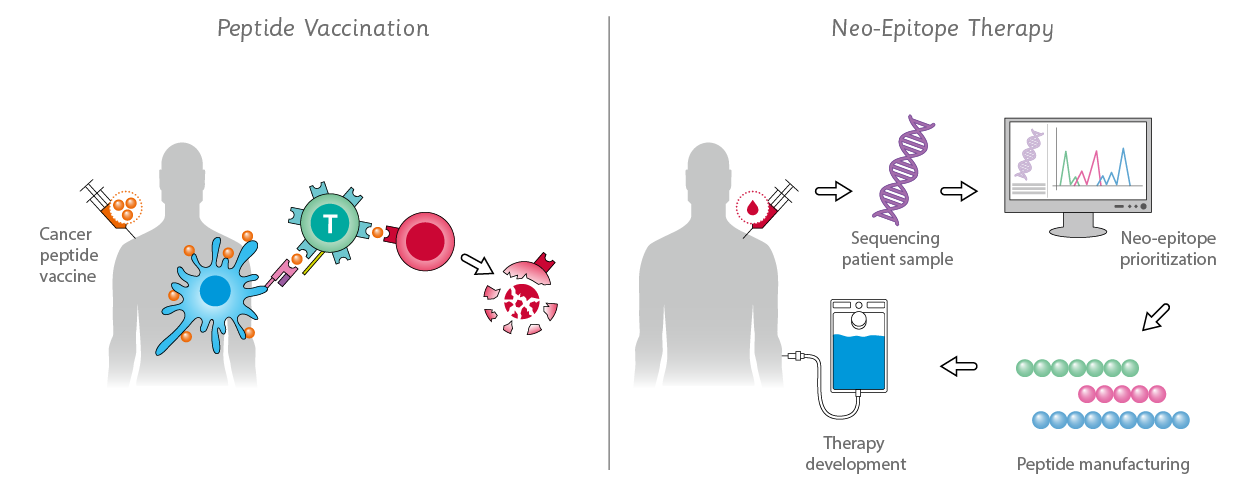
Neo-Epitope Therapy Development
Technical advances in high-throughput gene sequencing, bioinformatics and cancer immunotherapy enable the use of the host's immune system to treat cancer. Neo-epitopes can be recognized by the immune system to differentiate cancer from normal cells, making strong candidates for personalized cancer immunotherapy and neo-epitope vaccines. Our Clinical Peptides and Pools meet the need for high quality yet fast and affordable peptides for development of individualized therapies.
Why Work with JPT?
- Two decades worth of experience in peptide drug development and peptide chemistry
- Our algorithms (based on 2.5 million synthesized peptides) rank your neo-epitopes for manufacturability, stability, solubility, hydrophobicity and more
- Our efficent workflows result in fast turnaround, cost effectiveness, and success with long peptides
- Regulated according to ISO 9001:2015 standards
Clinical Peptides & Pools
JPT’s Clinical Peptides & Pools for development of immunotherapy & cell therapy are produced in production environments regulated by an enhanced ISO 9001:2015 quality management system for the stringent product requirements of immunotherapy such as peptide vaccines. Depending on the specifics of your immunotherapy protocols, the resulting products are applicable in clinical applications.
Please visit Clinical Peptides & Pools
PepMix™ Peptide Pools
Our antigen spanning PepMixes™ are available in a large variety of off-the-shelf peptide pools (e.g. for EBV, HCMV, Adenovirus, BKV, HHV6, and tumor associated antigens) or customized for your clinical application and antigens (e.g. epitope & neo-epitope, overlapping peptide pools and more). We also offer a wide variety of standardization and control pools such as CEFX Pools and HCMV (pp65).
Please visit PepMix™ Peptide Pools
Loading...