






PepStar™ Antigen Collection Pan-Coronavirus
Description
About PepStar™ Antigen Collection Pan-Coronavirus
Peptide microarray with peptides spanning the full proteome of Severe Acute Respiratory Syndrome-related coronavirus 2 (SARS-CoV-2) & Spike glycoprotein, Nucleoprotein, Envelope small membrane protein, Membrane protein of Coronaviruses Human SARS coronavirus (SARS-CoV), Middle East respiratory syndrome-related coronavirus (MERS), and common cold coronaviruses Human coronavirus 229E and OC43.
PepStar™ Antigen Collection Pan-Coronavirus - Specifications
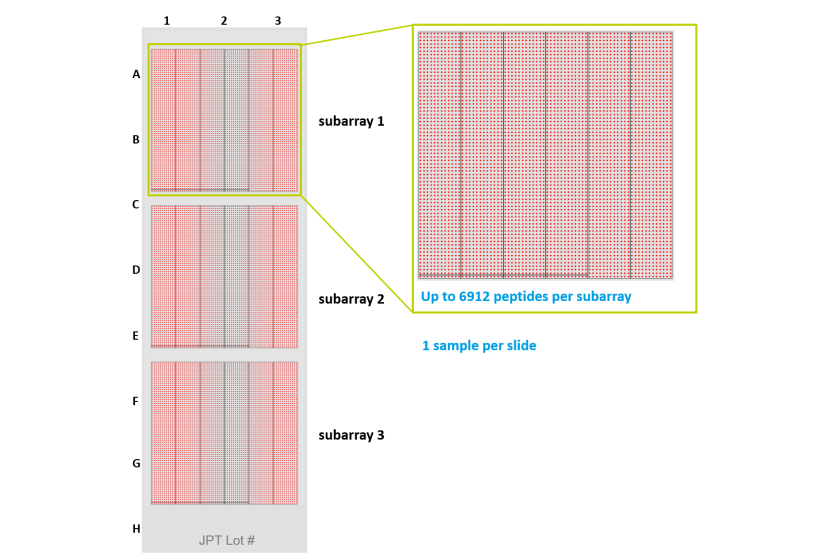
Number of peptides: 5828Amount: 1 microarray (1" x 3")
Delivery Format: 1-sample peptide microarray
Application(s): B-cell immunity
Condition(s)/Topic(s): Infection, Respiratory infection, Severe acute respiratory syndrome (SARS), Middle Eastern Respiratory Syndrome (MERS), Covid-19
Standard Delivery Time: approx. 10 days
Are you interested in your custom Peptide Microarrays? Choose your sequences, and array format. We will assist you along the way. Custom PepStar Peptide Microarrays.
PepStar Peptide Microarrays
JPT Peptide Technologies applies its unique peptide microarray platform to generate peptide microarrays on glass slides for biomarker discovery, immunomonitoring and detection and validation of protein interactions. Peptides are immobilized on glass slides via a flexible linker by chemoselective coupling. The covalently attached peptides are displayed infour copies on each slide. In addition, multiple identical copies of each microarray are produced. Incubation can be performed manually or automated using only minimal amounts of your sample. Read-out via fluorescence results in low background and high sensitivity.
Benefits of JPT's PepStar™ Peptide Microarrays
- Get hundreds of identical microarray copies
- Peptides are free of truncated sequences
- Small amounts of your precious samples are needed for incubation
- Applicable to a variety of biological samples (e.g. serum, plasma, antibody)
- Low background on glass surface
- Read-out via fluorescence
References
References for PepStar™ Antigen Collection Pan-Coronavirus
References:
Read References with PepStar & RepliTope Peptide Microarrays & COVID-19 related publications
Application Notes
A Modular Approach for Epitope Discovery and High-Resolution Profiling of Humoral Immune Responses
Pawlowski et al. (2013) Full text
Multiple Sclerosis and Epstein Barr Virus Infection. An Epitope Mapping Study
Reimer et al. (2014) Full text
The Challenge of Antigen Sequence Diversity:Solutions with ULTRA Peptide Libraries
Pawlowski et al. (2015) Full text
Rapid Mimotope Optimization for Pharmacokinetic Analysis of the Novel Therapeutic antibody IMAB362
Daneschar et al. (2014) Full text
Testimonials for JPT's Peptide Microarrays
"We at CNAM have studied the immune response of infected or control patients (Covid-19, asymptomatic, or pre-epidemic sera) by epitope mapping against peptides derived from the structural peptides of SARs-CoV-2. For our antibody mapping studies we have used the JPT's PepStar peptide microarrays and their comprehensive microarray profiling services. PepStar peptide microarrays with their very high quality and batch-to-batch reproducibility in combination with easy read-out and JPT’s personalized customer support have provided excellent results. We are very pleased to have worked with the JPT technology and plan to work again with them in the future.."
Prof. Jean-Francois Zagury, Conservatoire National des Arts et Métiers, Paris, France
"We utilize JPT's multiwell microarray assay service to study autoimmune diseases such as rheumatoid arthritis. Our laboratory focuses on the mechanisms by which rheumatoid arthritis-associated HLA-DR molecules contribute to the development of anti-citrullinated protein antibodies (ACPAs) in rheumatoid arthritis. We have developed a sensitive peptide array assay, capable to detect and monitor ACPAs in mice (Front Immunol, 2022). JPT’s multiwell microarray assay service based on their PepStar peptide microarrays has been a most reliable and robust approach for our experiments. Their full profiling and data interpretation service with its excellent documentation has helped to further our research of rheumatoid arthritis.."
Dr. Isabelle Auger, INSERM UMRs 1097, Marseille, France
"We study modulation of dopaminergic neurotransmisson by TAAR1 and D2R. The functional interaction of the two receptors in the brain supports TAAR1 as a target for the treatment of psychiatric disorders such as schizophrenia or bipolar disorder. To map the epitopes of our newly generated specific anti-rat TAAR1 antibodies we used JPT's PepStarTM peptide microarrays. The peptide microarrays greatly contributed to our successful and recently published study. We were very satisified with the exceptional product and service delivered by JPT Peptide Technologies as well as their scientific Customer Support which was always at our disposal."
Stefan Obermüller, F.Hoffmann - La Roche Ltd., Roche Pharma Research and Early Development, Basel, Switzerland
Documentation
Documentation for PepStar™ Antigen Collection Pan-Coronavirus
Properties
Properties of PepStar™ Antigen Collection Pan-Coronavirus
| Properties | Values |
|---|---|
| Amount: | 1 microarray (1" x 3") |
| Application: | B-cell Immunity |
| Category: | PepStar Peptide Microarrays |
| Condition / Topic: | Covid-19, Infection, Middle Eastern Respiratory Syndrome (MERS), Respiratory infection, Severe acute respiratory syndrome (SARS) |
| Layout: | 1-sample peptide microarray |
| Organism: | Human coronavirus (HCoV), MERS-CoV (Middle East respiratory syndrome-related coronavirus), SARS-CoV (Severe acute respiratory syndrome coronavirus), SARS-CoV-2 (Severe Acute Respiratory Syndrome-related coronavirus 2) |
| Protein Name: | Selected proteins |
| Quantification: | Yes |


Quality Assurance
All production is ISO 9001:2015 certified.


.jpg)
.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)

.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)
-Peptide-Pool.jpg)